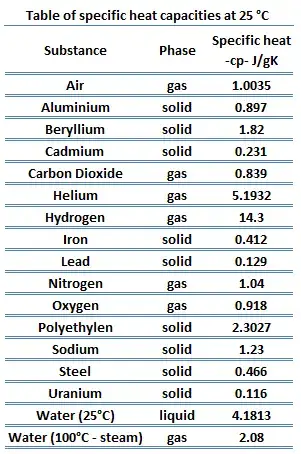
Derived mean values of the specific heat of pure iron in comparison... | Download Scientific Diagram

6pc Specific Heat Metal Cylinders Set - Copper, Lead, Brass, Zinc, Iron & Aluminum - Includes Wooden Storage Block - for Specific Heat, Specific Gravity & Density Experimentation - Eisco Labs: Amazon.com:
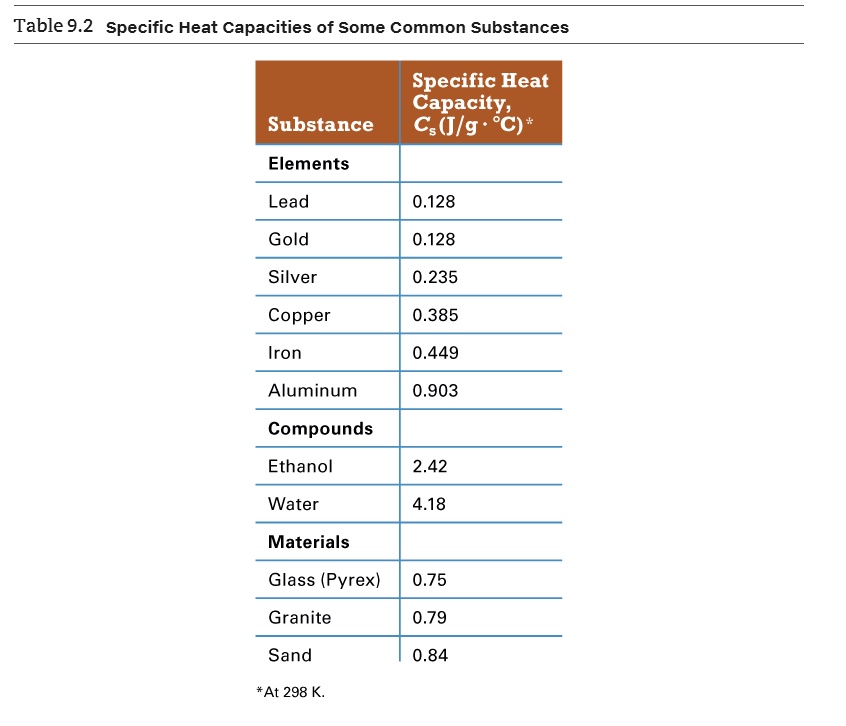
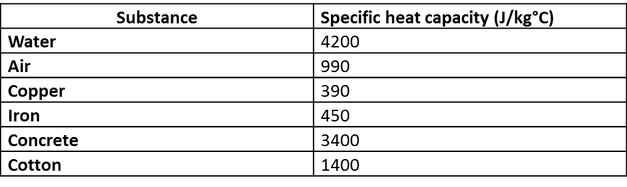
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42
![PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/36a34a8a61709b713d9e6e7e12f0b93504140c9c/7-Figure2-1.png)
PDF] Electronic Specific Heat of Iron Pnictides Based on Electron-Cooper Pair Interaction | Semantic Scholar

Composition and structure dependence of specific heat of disordered iron-palladium alloys - ScienceDirect
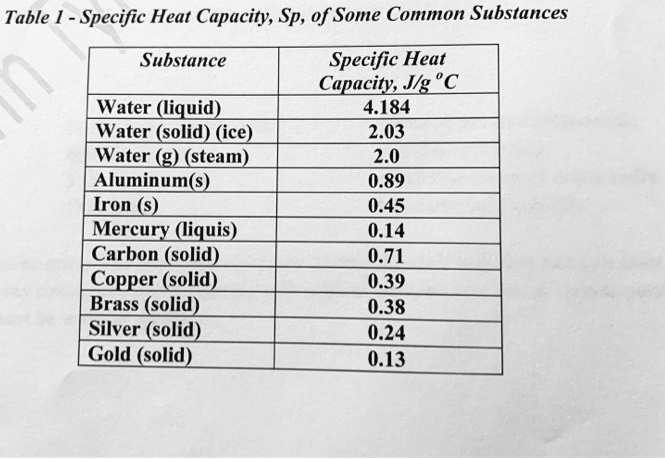
SOLVED: Table 1 Specific Heat Capacity, Sp, of Some Common Substances Substance Specific Heat Capacity, Jg 4.184 2.03 2.0 0.89 0.45 0.14 0.71 0.39 0.38 0.24 0.13 Water (liquid) Water (solid) (ice)
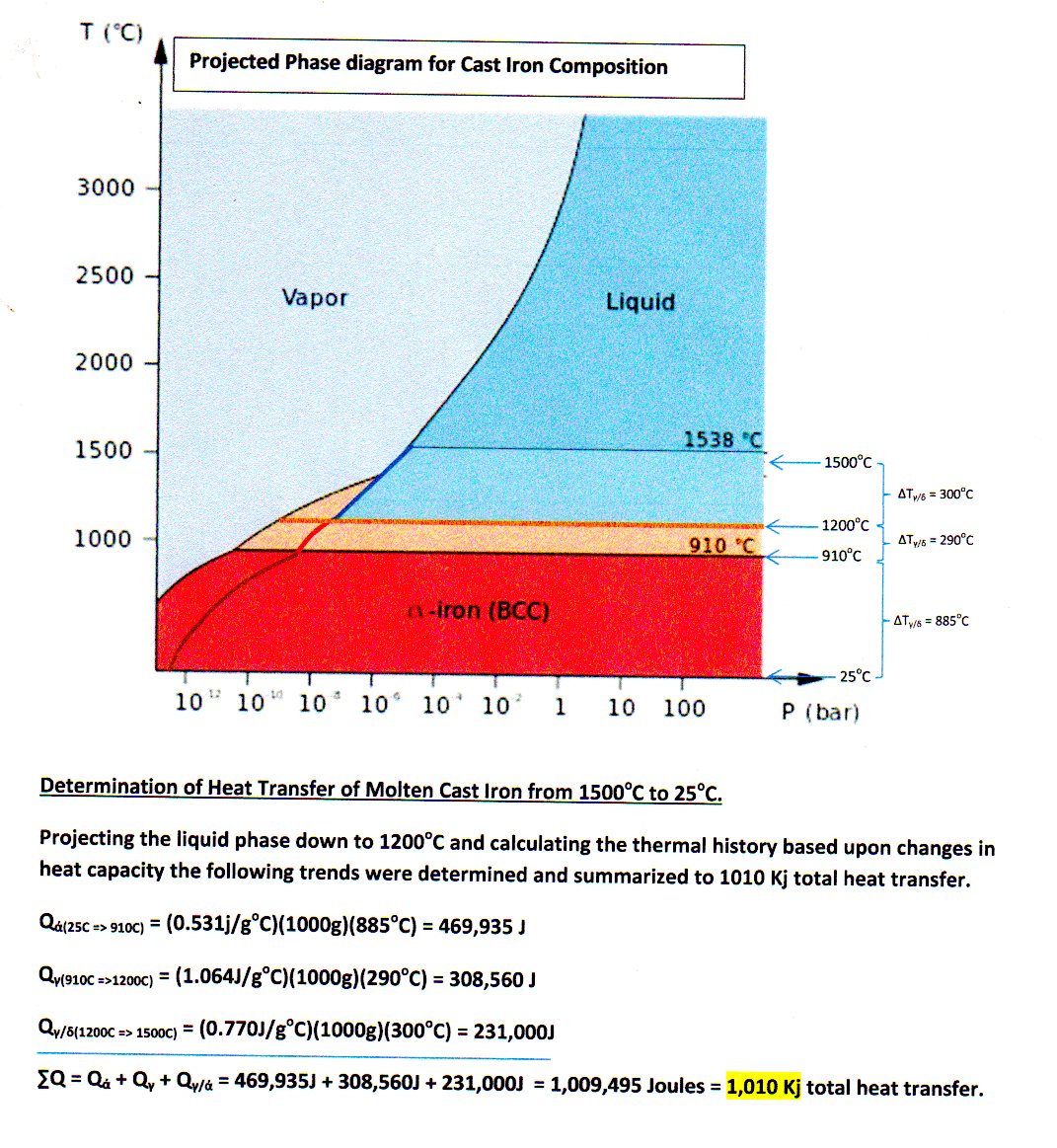
Molten iron is extremely hot, averaging about 1,500 C. The specific heat of iron is 0.46 J/gC. How much heat is released to the atmosphere when 1 kg molten iron cools to
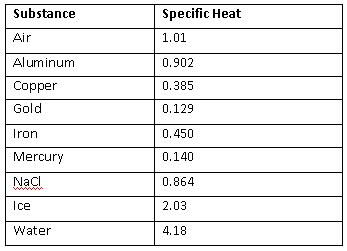
A 36.07 g sample of a substance is initially at 27.8°C. After absorbing 2639 J of heat, the temperature of the substance is 109.0°C. What is the specific heat of the substance? | Socratic
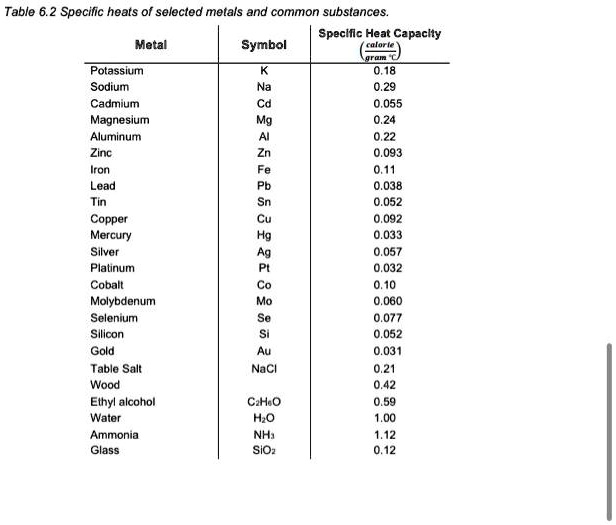



![Q. 3.37 If the same amount of heat is su... [FREE SOLUTION] | StudySmarter Q. 3.37 If the same amount of heat is su... [FREE SOLUTION] | StudySmarter](https://studysmarter-mediafiles.s3.amazonaws.com/media/textbook-exercise-images/image_fqdOuye.png?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIA4OLDUDE42UZHAIET%2F20230619%2Feu-central-1%2Fs3%2Faws4_request&X-Amz-Date=20230619T113149Z&X-Amz-Expires=90000&X-Amz-SignedHeaders=host&X-Amz-Signature=9addeb669c18772e2f62a5496f76a61c0c41bc11c2f7d3c5d66f3d6f112adfcc)
![ANSWERED] Metal Specific Heat (J/g°C) Calcium 0.647 ... - Organic Chemistry ANSWERED] Metal Specific Heat (J/g°C) Calcium 0.647 ... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/75254282-1659635207.98119.jpeg)