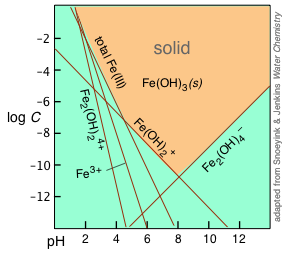
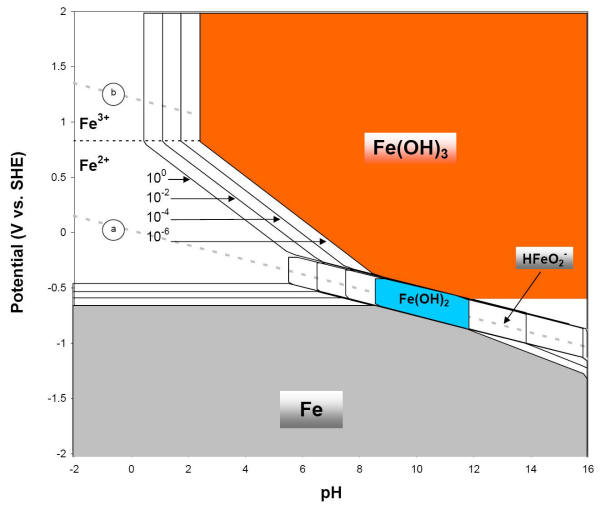
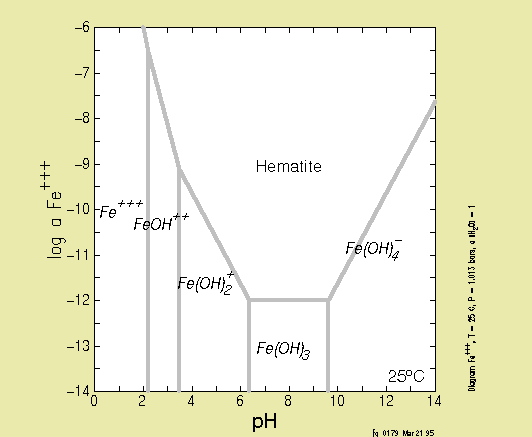
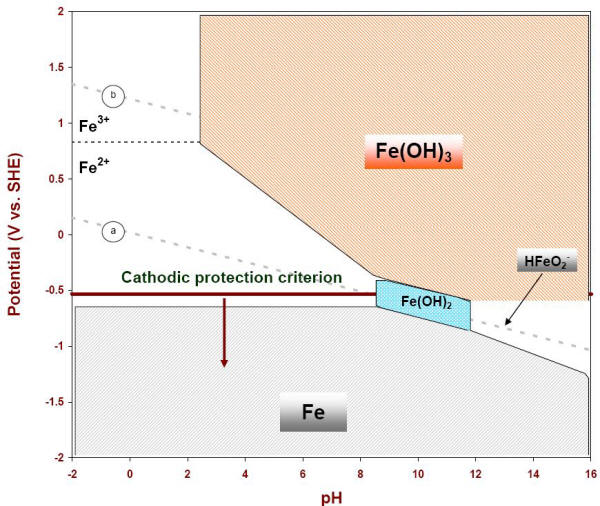
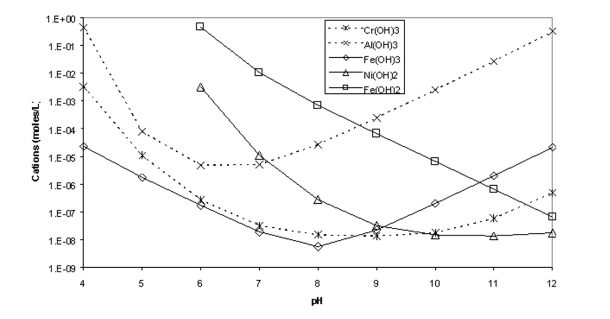
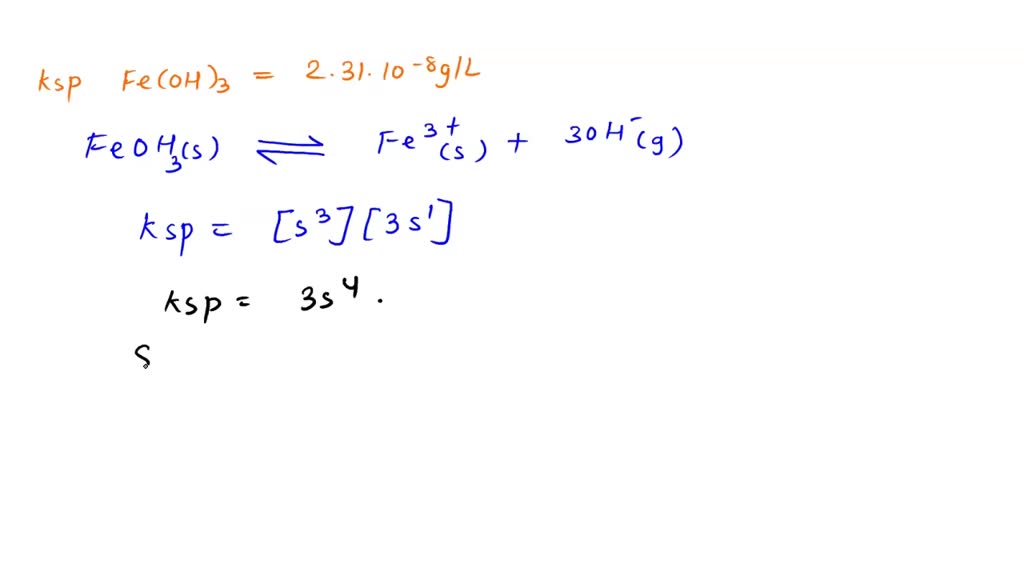Solubility Model for Ferrous Iron Hydroxide, Hibbingite, Siderite, and Chukanovite in High Saline Solutions of Sodium Chloride, Sodium Sulfate, and Sodium Carbonate | ACS Earth and Space Chemistry

SOLVED: The solubility of iron(II) hydroxide, Fe(OH)2, is 1.43x10-3-gram per liter at 25oC. (a) Write a balanced equation for the solubility equilibrium. (b) Write the expression for the solubility product constant, Ksp,
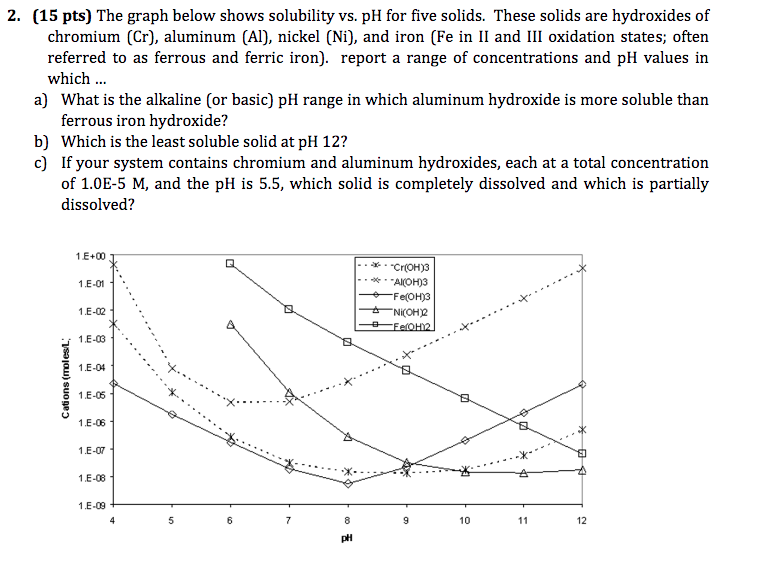
The solubility of iron (II) hydroxide, Fe(OH)2, is 1.43 x10^–3 g/L. Calculate the Ksp for iron (II) hydroxide. - Sarthaks eConnect | Largest Online Education Community
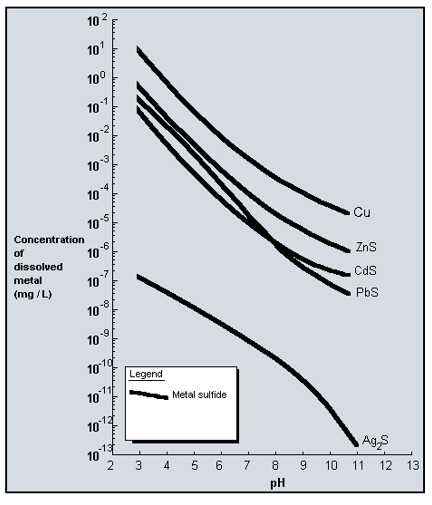
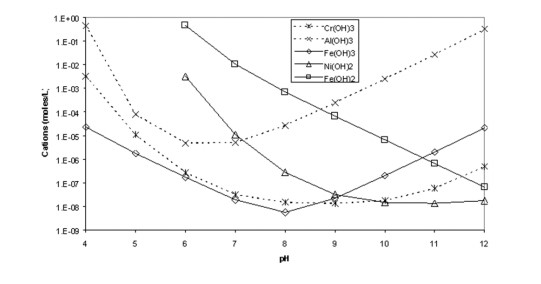
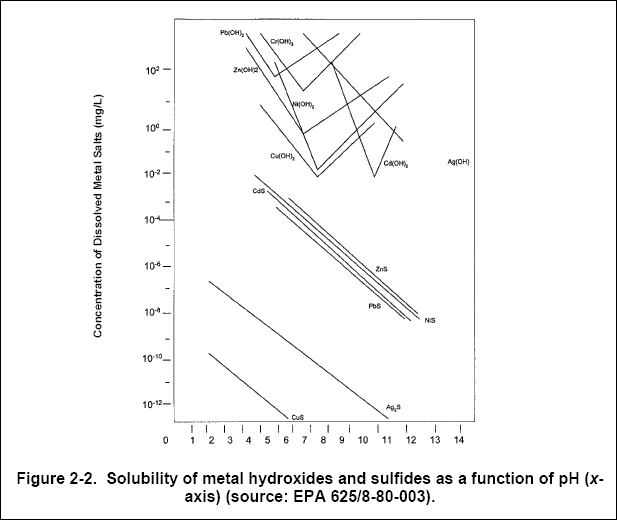
Hydroxide Precipitation of Metals | Hoffland Environmental, Inc | (936) 856.4515 - Manufacturer of Industrial Wastewater Treatment Systems

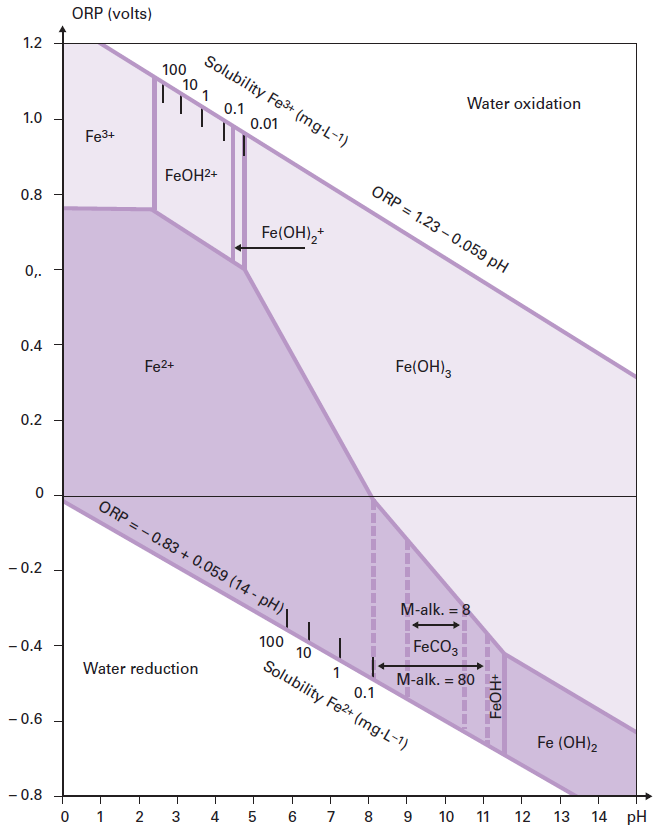
Solubility of Fe(II) vs pH, as controlled by the solubility of Fe(OH) 2... | Download Scientific Diagram

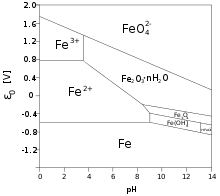
The calculation of the solubility of metal hydroxides, oxide-hydroxides, and oxides, and their visualisation in logarithmic diagrams | SpringerLink