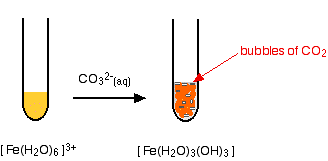
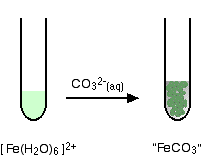
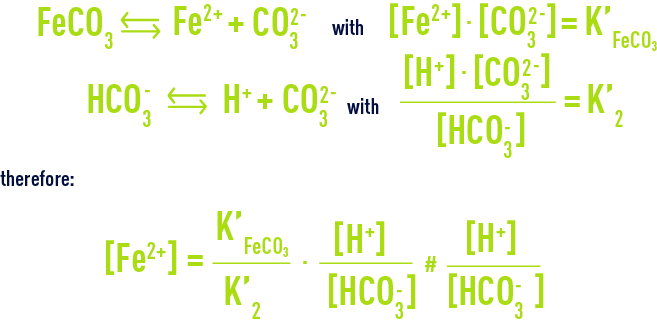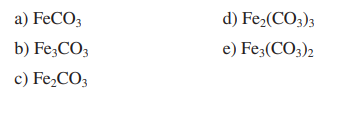Fe(III) mobilisation by carbonate in low temperature environments: Study of the solubility of ferrihydrite in carbonate media and the formation of Fe( III) carbonate complexes - ScienceDirect
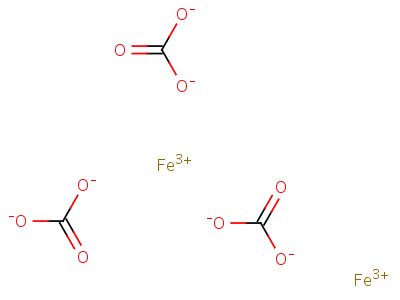
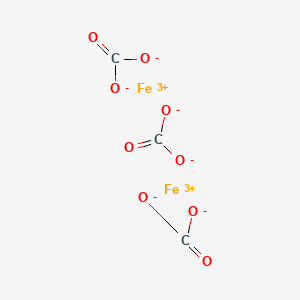
10290-71-8;26273-46-1;58499-83-5, iron(3+) carbonate (2:3), CAS No 10290-71-8;26273-46-1;58499-83-5 iron(3+) carbonate (2:3)