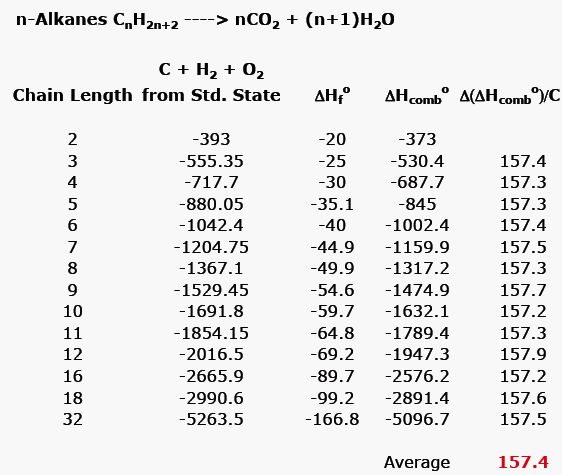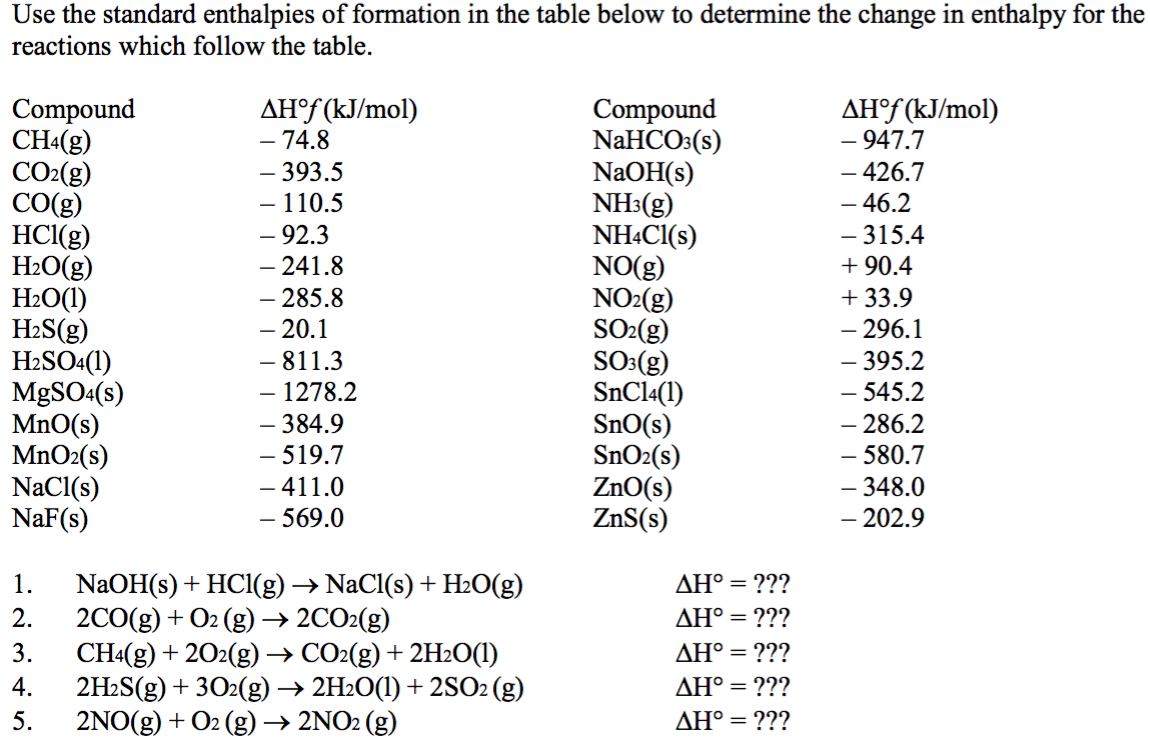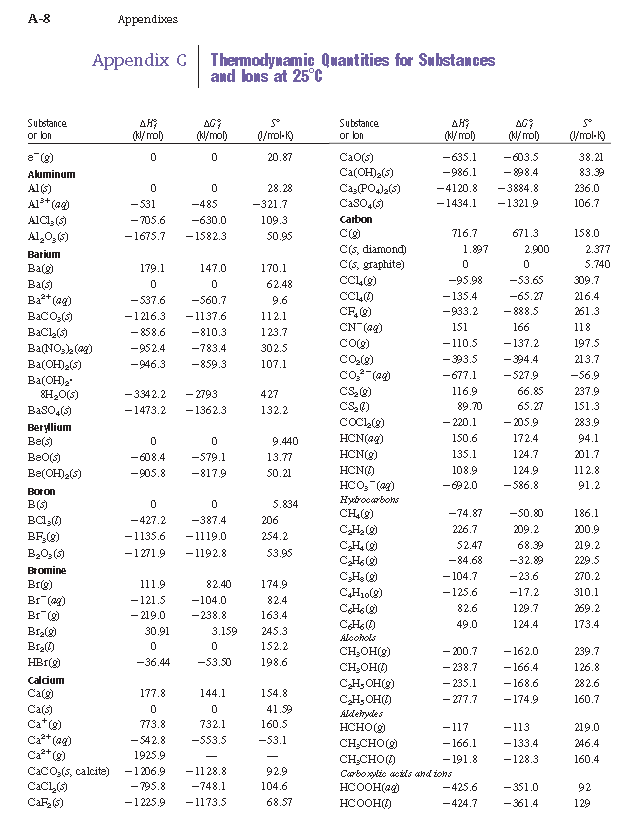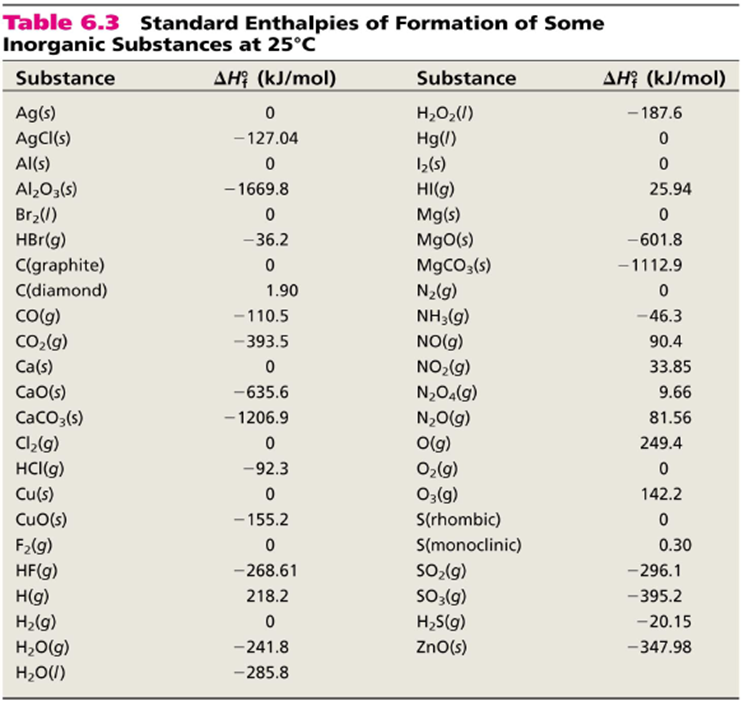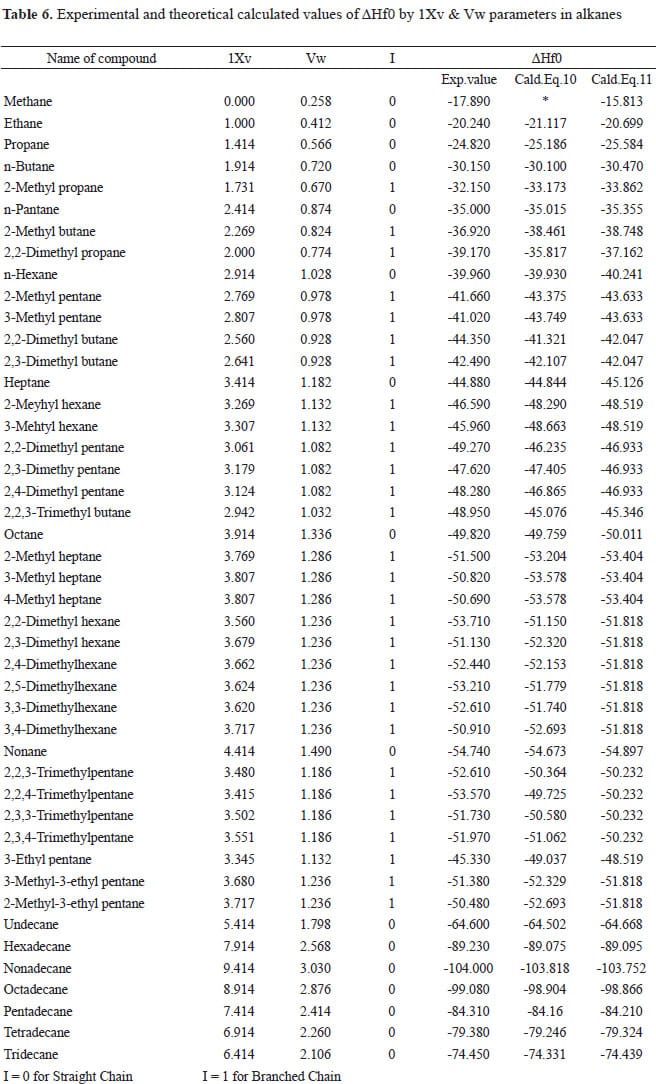
Which of the enthalpies of combustion in Table 5.2 the table are also standard enthalpies of formation? | bartleby
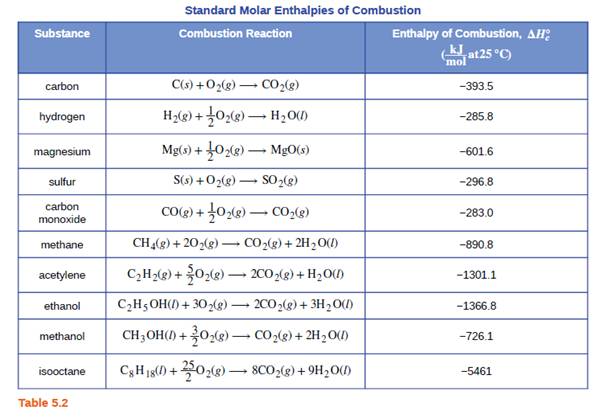
STANDARD HEAT OF FORMATION. DEFINITION The change in enthalpy that accompanies the formation of one mole of the compound at its normal state from its. - ppt download
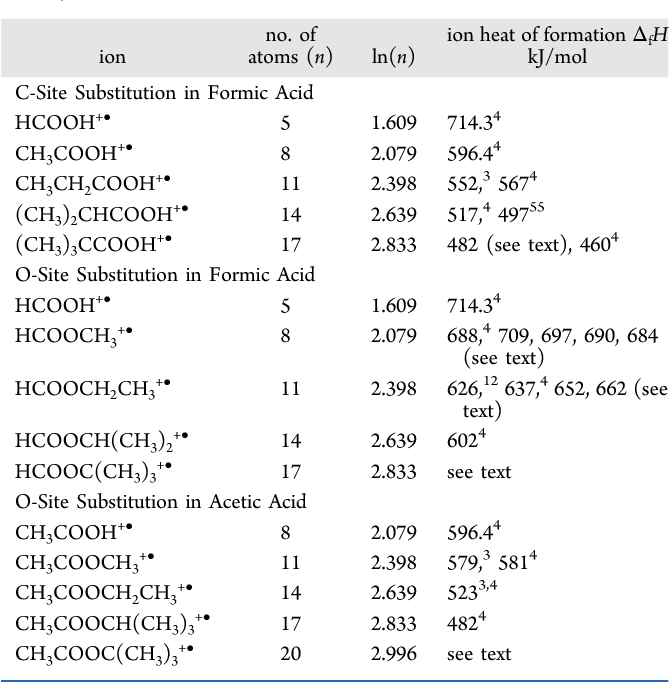
Table 7 from Size effects on cation heats of formation. II. Methyl substitutions in oxygen compounds. | Semantic Scholar
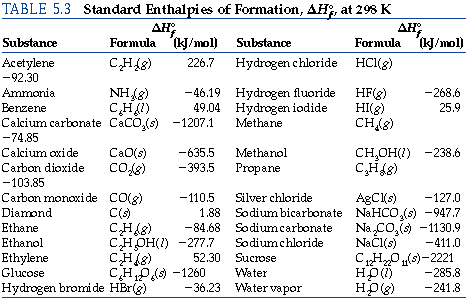
Calculate the enthalpy of reaction for the reaction "CH"_3"COOH" + "H"_2"O" -> "CH"_3"CH"_2"OH" + "O"_2? | Socratic

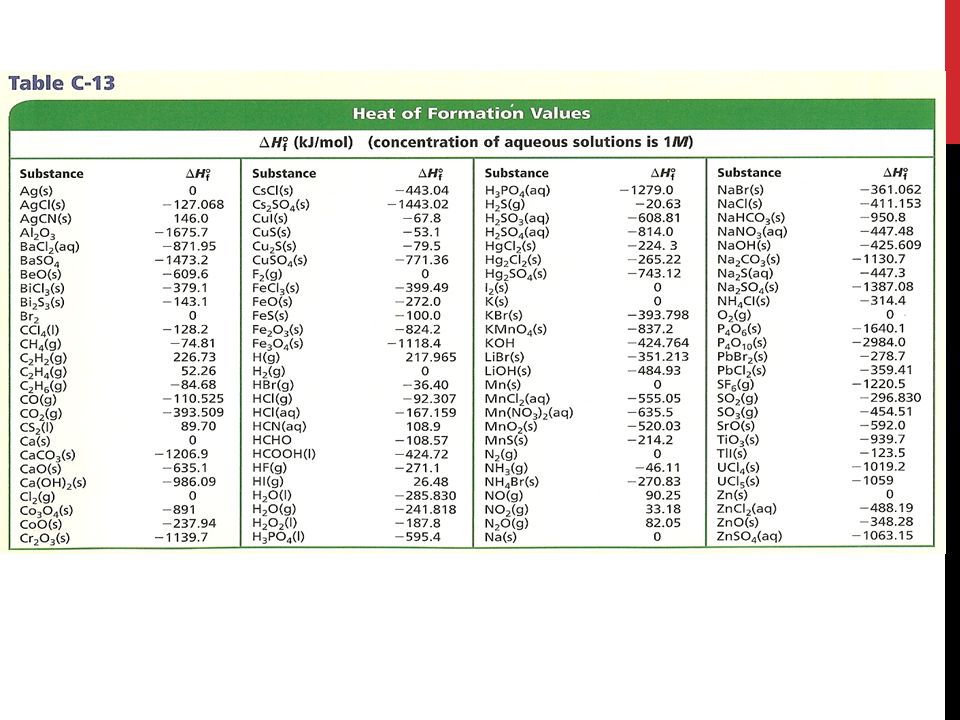
physical chemistry - How do you calculate the heat of a reaction given a table of heat of formation values? - Chemistry Stack Exchange
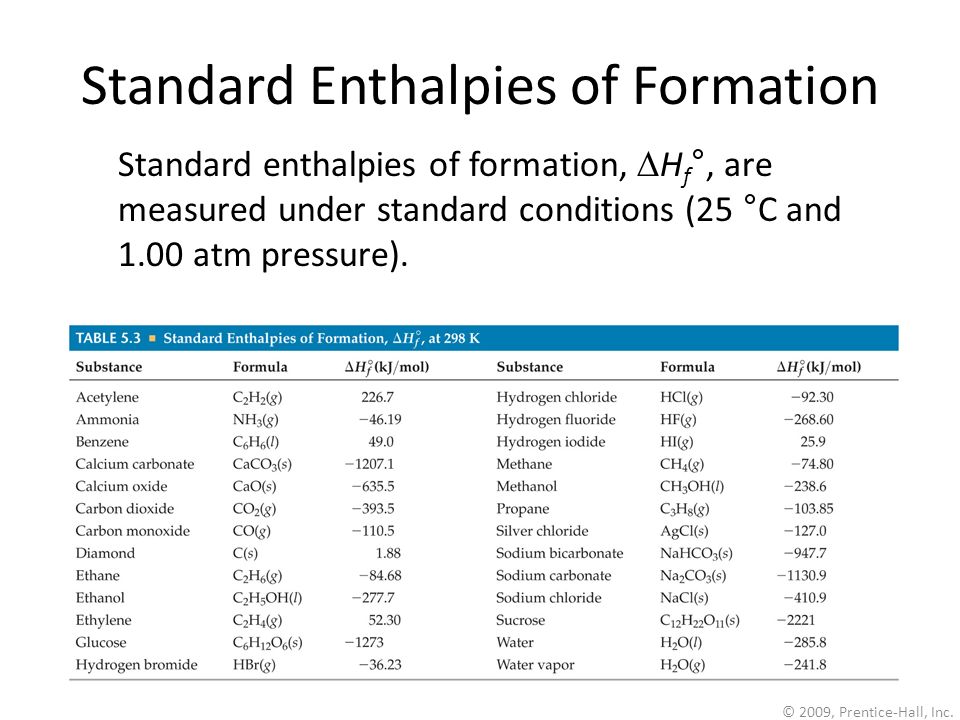
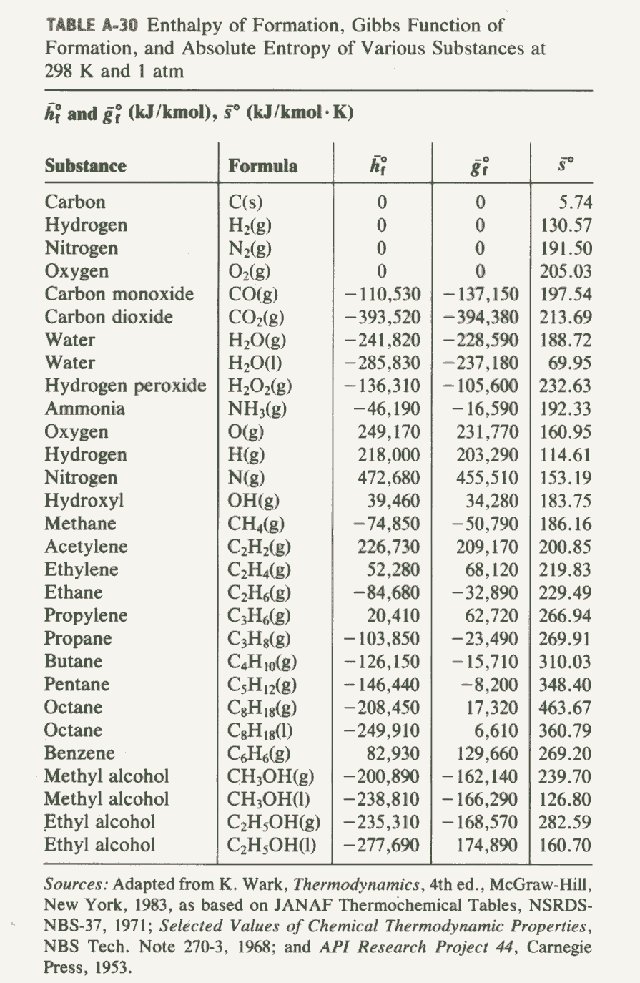
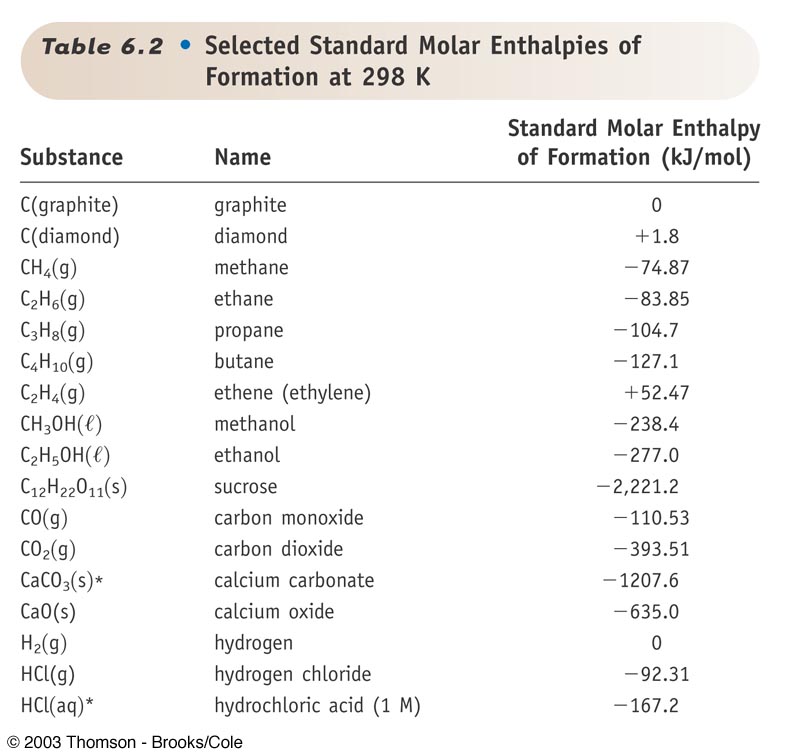
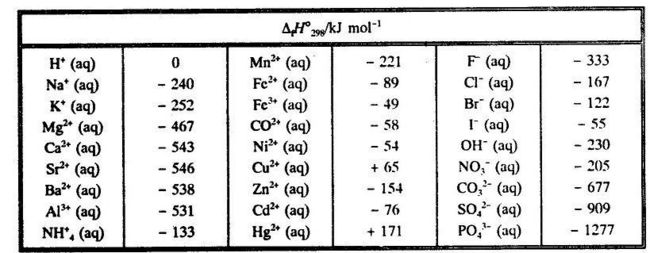


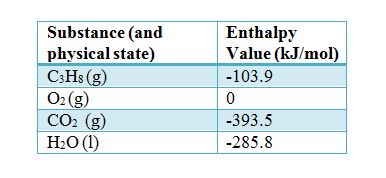
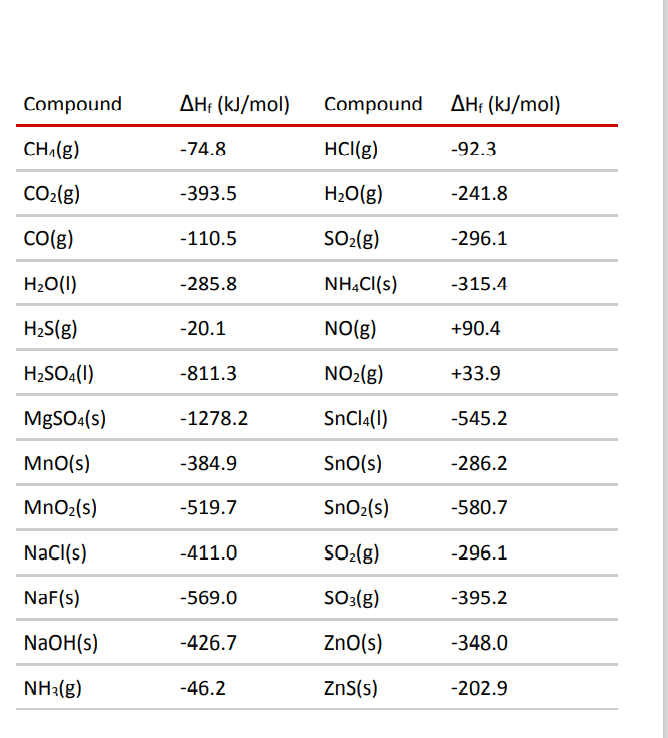



:max_bytes(150000):strip_icc()/GettyImages-154953454-6806780a2f0f4ec99daf580619b5aeef.jpg)